Enabling Biomarker-Driven Diagnostics from Discovery to Deployment
Companion diagnostics (CDx) are at the core of precision medicine, enabling the identification of patients most likely to benefit from targeted therapies. At Creative Enzymes, we provide a comprehensive CDx enzyme platform that integrates enzyme products, engineering technologies, and assay development capabilities to support the full lifecycle of diagnostic innovation.
Our platform empowers pharmaceutical companies, diagnostic developers, and research institutions to accelerate biomarker-driven decision-making—transforming molecular insights into clinically actionable diagnostic solutions.
Companion diagnostics are in vitro diagnostic tools designed to determine the suitability of specific therapeutic interventions based on individual patient biomarkers.
Improve treatment efficacy through patient stratification.
Reduce clinical trial risks and failure rates.
Enable targeted therapy selection.
Support regulatory approval of precision therapeutics.
As precision medicine continues to evolve, CDx development requires highly reliable biochemical components—especially enzymes that drive signal detection, amplification, and assay performance.
Enzymes are the functional core of many diagnostic systems. Their catalytic properties enable highly sensitive, rapid, and specific detection of biological signals, making them indispensable in companion diagnostic (CDx) applications across immunoassays, molecular diagnostics, and biosensor platforms. By converting molecular recognition events into measurable outputs, enzymes bridge the gap between biological interactions and analytical readouts.
-systems.webp)
Enzymes catalyze reactions that produce measurable outputs such as colorimetric, fluorescent, or chemiluminescent signals.
In CDx assays, enzymes such as HRP and ALP are widely used to convert non-detectable substrates into visible or quantifiable signals. These reactions form the basis of widely adopted detection technologies including ELISA, CLIA, and fluorescence-based assays. The efficiency and kinetics of these enzymatic reactions directly influence signal intensity, detection accuracy, and assay reproducibility.
Through catalytic turnover, enzymes amplify weak biological signals, enabling detection of low-abundance biomarkers.
A single enzyme molecule can catalyze multiple reaction cycles, significantly enhancing signal output from minimal target presence. This amplification is essential for detecting low-copy-number nucleic acids, rare proteins, or early-stage disease biomarkers. Enzyme-driven amplification mechanisms are fundamental to technologies such as PCR/qPCR, enzyme-linked immunoassays, and cascade amplification systems used in advanced biosensors.
Engineered enzymes enhance assay specificity by minimizing background noise and improving target selectivity.
High-purity and optimized enzymes reduce non-specific reactions and improve signal-to-noise ratios, which is critical for accurate clinical interpretation. Additionally, enzyme conjugation strategies (e.g., antibody-enzyme labeling) and substrate selectivity further enhance assay precision. Advanced enzyme engineering enables fine-tuning of catalytic activity and substrate affinity, ensuring reliable performance even in complex biological samples.
Our CDx enzyme portfolio supports immunoassays, molecular diagnostics, and NGS workflows, providing a scalable foundation for companion diagnostic development.
| Catalog | Product Name |
|---|---|
| ENZD-P01 | Glyceryl-free Heat-labile UDG enzyme |
| ENZD-P02 | Taq HS DNA Polymerase |
| Catalog | Product Name |
|---|---|
| ENZD-P02 | Taq HS DNA Polymerase |
| ENZD-P03 | EnzSmart U+ Taq HS DNA Polymerase |
| ENZD-P04 | AdvTaq DNA Polymerase |
| ENZD-P05 | EnzSmart U+ Melt Pro Multiplex DNA Polymerase |
| ENZD-P06 | EnzSmart MethyLight DNA Polymerase |
| ENZD-P09 | Bst II Pro DNA Polymerase Large Fragment |
| ENZD-P10 | Bst DNA Polymerase Large Fragment |
| Catalog | Product Name |
|---|---|
| ENZD-MDLyo01 | AdvSTART Reverse Transcriptase(Glycerol-free) |
| ENZD-MDLyo02 | High Reverse Transcriptase II (Glycerol-free) |
| ENZD-MDLyo03 | Murine RNase Inhibitor (Glycerol-free) |
| ENZD-RT01 | Murine RNase Inhibitor Pro |
| ENZD-RT02 | AdvSTART Reverse Transcriptase |
| ENZD-RT03 | High Reverse Transcriptase II |
| ENZD-RT04 | High Reverse Transcriptase III |
| ENZD-RT05 | Murine RNase inhibitor |
These systems are suited for routine quantitative testing, infectious disease detection, SNP analysis, and biomarker-driven diagnostic assay development. Optimized mixes support strong fluorescence performance, low background, robust multiplex capability, and streamlined workflow integration for highly reliable nucleic acid analysis.
| Catalog | Product Name |
|---|---|
| ENZD-QM01 | UltiPrime U+ All-Powerful Multiple Probe qPCR PreMix (Low DNA) |
| ENZD-QM02 | UltiPrime U+ Super Multiple Probe qPCR PreMix (Low DNA) |
| ENZD-QM03 | Universal U+ Probe Master Mix V2 |
| ENZD-QM04 | EnzSmart U+ All-Powerful Multiple Probe qPCR PreMix (ONE TUBE) |
| ENZD-QM05 | EnzSmart U+ Super Multiple Probe qPCR PreMix (ONE TUBE) |
| ENZD-QM06 | Taq Pro U+ Multiple Probe qPCR Mix |
| ENZD-QM07 | Taq Pro HighGC U+ Multiple Probe qPCR Mix |
| ENZD-QM08 | Hot-start Geno-SNP Probe Master Mix |
| ENZD-QM09 | qPCR Probe Master Mix |
| ENZD-QM10 | U+ Probe Master Mix |
| ENZD-QM11 | Animal Detection U+ Probe qPCR Super PreMix |
| ENZD-QM12 | EnzSmart MethyLight qPCR Mix |
| Catalog | Product Name |
|---|---|
| ENZD-RT08 | ColorDetect LAMP/RT-LAMP Mix |
| ENZD-RT09 | Fluorescent LAMP/RT-LAMP Kit |
These reagents support high-quality library construction, target enrichment, sequencing compatibility, and reliable amplification in genomics-driven CDx workflows. They are particularly relevant for mutation profiling, liquid biopsy development, transcriptome analysis, and advanced molecular characterization in precision medicine programs.
| Catalog | Product Name |
|---|---|
| DIA-554 | HiFi Amplification Mix |
| DIA-555 | HiFi Universal Amplification Mix for Illumina |
| DIA-556 | HiFi Universal Amplification Mix for MGI (SI) |
| DIA-557 | mRNA Capture Beads |
| DIA-558 | mRNA Capture Beads 2.0 |
| DIA-559 | Globin mRNA Depletion Probe for Prevalent Species |
| DIA-560 | High-Fidelity Amplification Mix |
| DIA-561 | ds-cDNA Synthesis Module (+gDNA wiper) |
| DIA-562 | Pathogen Multiplex PCR Mix for DNA |
| DIA-563 | Pathogen Multiplex PCR Mix for DNA&RNA |
| DIA-564 | Broad-Spectrum Adapter Ligation Module, Illumina-Compatible |
| DIA-565 | Broad-Spectrum End Preparation Module, Illumina-Compatible, Version II |
| DIA-582 | DNA Polymerase I Klenow Fragment |
| DIA-583 | DNA Polymerase I Klenow Fragment exo- |
| DIA-584 | T4 DNA Ligase (Rapid) |
| DIA-585 | T4 DNA Polymerase |
| DIA-586 | T4 Polynucleotide Kinase |
| DIA-587 | Amplify Enzyme |
These tools improve sample readiness for downstream sequencing and transcriptional analysis by reducing unwanted background RNA and enriching meaningful targets. They are useful in expression-based biomarker discovery, circulating nucleic acid analysis, and diagnostic workflows where clean RNA input is essential for accurate interpretation.
| Catalog | Product Name |
|---|---|
| DIA-557 | mRNA Capture Beads |
| DIA-558 | mRNA Capture Beads 2.0 |
| DIA-559 | Globin mRNA Depletion Probe for Prevalent Species |
| Kit-032 | FastSelect rRNA Kit (Human) |
| Kit-033 | rRNA Depletion Kit for Arthropoda/Insects/Parasites |
| Kit-034 | rRNA Depletion Kit for Aquatic Animals |
| Kit-035 | rRNA Depletion Kit for Animals |
| Kit-036 | rRNA Depletion Kit for Bacteria |
| Kit-037 | rRNA Depletion Kit for Fungi |
| Kit-038 | rRNA Depletion Kit for Model Species |
| Kit-039 | rRNA Depletion Kit for Poultry/Birds |
| Kit-040 | rRNA Depletion Kit for Plant |
| Kit-041 | rRNA Depletion Kit for Prevalent Species |
| Kit-042 | rRNA Depletion Kit for Reptiles/Mammals |
| Development Focus | Description |
|---|---|
| Activity Optimization | Enhancement of catalytic efficiency and functional performance for assay-specific signal output. |
| Stability Engineering | Improvement of thermal tolerance, storage resilience, and compatibility with lyophilized or glycerol-free formulations. |
| Application Tailoring | Customized enzyme design to fit specific diagnostic chemistries, detection systems, and workflow requirements. |
.webp)
We offer advanced enzyme engineering services to meet the stringent performance and reliability requirements of companion diagnostic (CDx) applications. By integrating protein engineering, process optimization, and rigorous quality control, we ensure enzymes are tailored for high-sensitivity diagnostic workflows.
Removal of host cell proteins, residual DNA, and process-related impurities to minimize background interference and reduce false-positive or false-negative results in diagnostic assays.
Optimization for thermal resistance, long-term storage, and lyophilization compatibility—ensuring consistent enzyme performance across transportation, storage, and point-of-care testing (POCT) conditions.
Enhancement of catalytic efficiency and substrate affinity to improve assay sensitivity, lower detection limits, and expand dynamic range for low-abundance biomarker detection.
Strict quality control and process standardization to ensure high lot-to-lot reproducibility, supporting reliable assay performance in both research and large-scale diagnostic production.
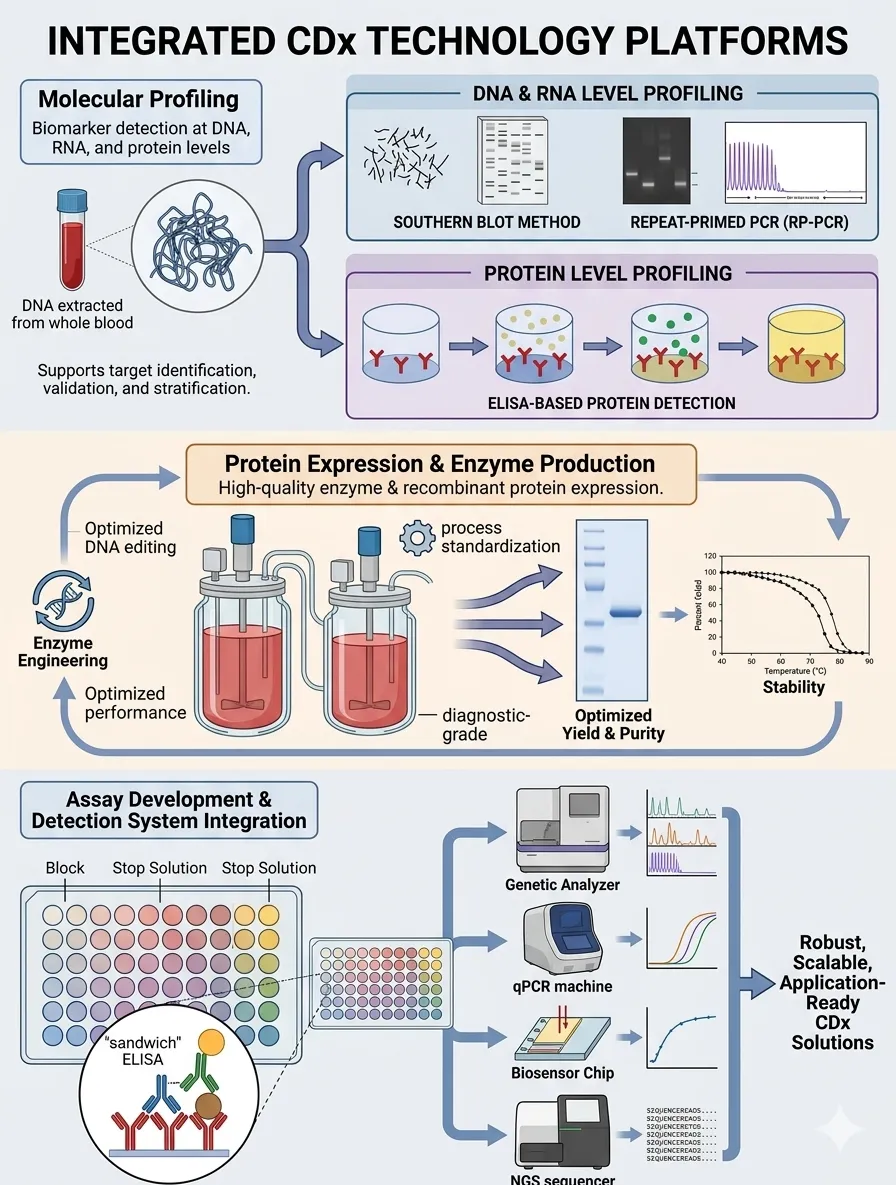
Our integrated technology platforms support the entire CDx development pipeline, from early-stage biomarker discovery to assay development and system integration. By combining multi-level capabilities across molecular biology, protein engineering, and diagnostic technologies, we enable efficient and scalable CDx development.
Biomarker detection at DNA, RNA, and protein levels using advanced molecular and immunological approaches, supporting target identification, validation, and stratification in precision medicine.
High-quality enzyme production and recombinant protein expression tailored for diagnostic applications, with optimized yield, purity, and functional performance.
Design, development, and analytical validation of diagnostic assays with improved sensitivity, specificity, and reproducibility across different platforms and sample types.
Support for integration into diverse diagnostic formats, including ELISA, PCR/qPCR, biosensors, and next-generation sequencing (NGS)-based systems.
Our platforms enable seamless transition from biomarker discovery to robust, scalable, and application-ready CDx solutions.
We provide end-to-end support for companion diagnostic (CDx) development, covering key stages from biomarker translation to assay optimization and scalable implementation. Our integrated approach ensures efficient development while maintaining high analytical performance and reliability.
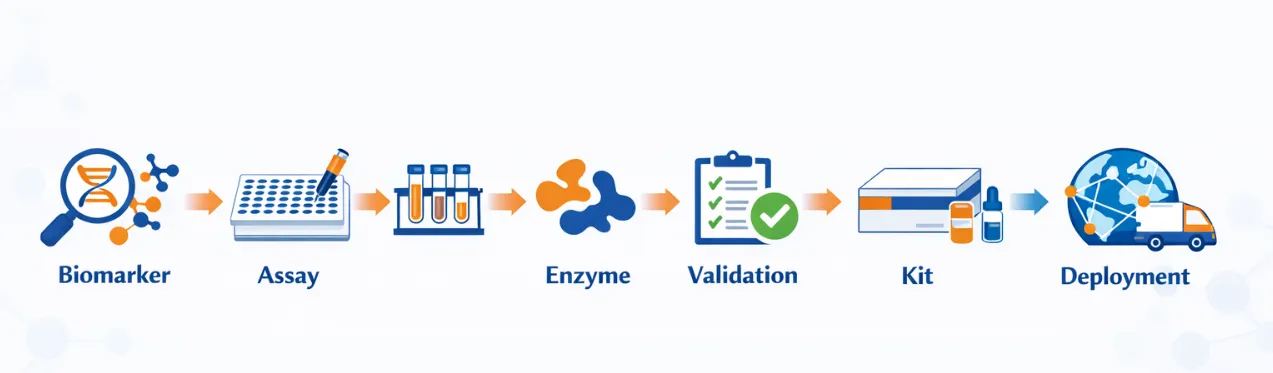
Our solutions are tailored to accelerate development timelines while reducing technical risk, enabling seamless transition from research to application-ready CDx systems.
To demonstrate the practical value of our CDx enzyme platform, we developed two representative nucleic acid detection workflows based on optimized enzyme systems: a probe-based qPCR assay for high-sensitivity quantitative detection and a LAMP assay for rapid visual and fluorescence readout.
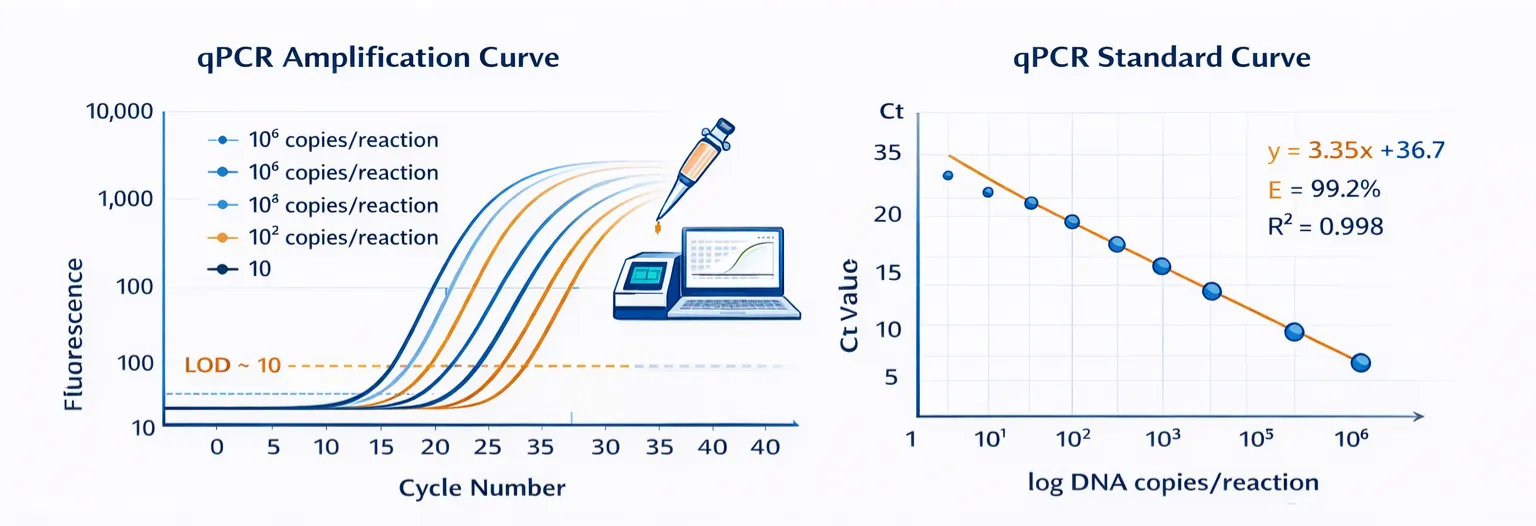
In the qPCR workflow, an optimized polymerase system was used to detect a serial dilution of target nucleic acid templates across a wide dynamic range. The amplification plot showed clear separation among different input concentrations, with high-copy samples reaching the fluorescence threshold earlier and low-copy samples amplifying later, consistent with expected qPCR kinetics. The system achieved a theoretical limit of detection of approximately 10 copies per reaction.
The corresponding standard curve demonstrated excellent linearity between Ct value and template concentration, with an amplification efficiency of 99.2% and an R² value of 0.998. These results suggest that the enzyme system is suitable for both sensitive detection and quantitative analysis.
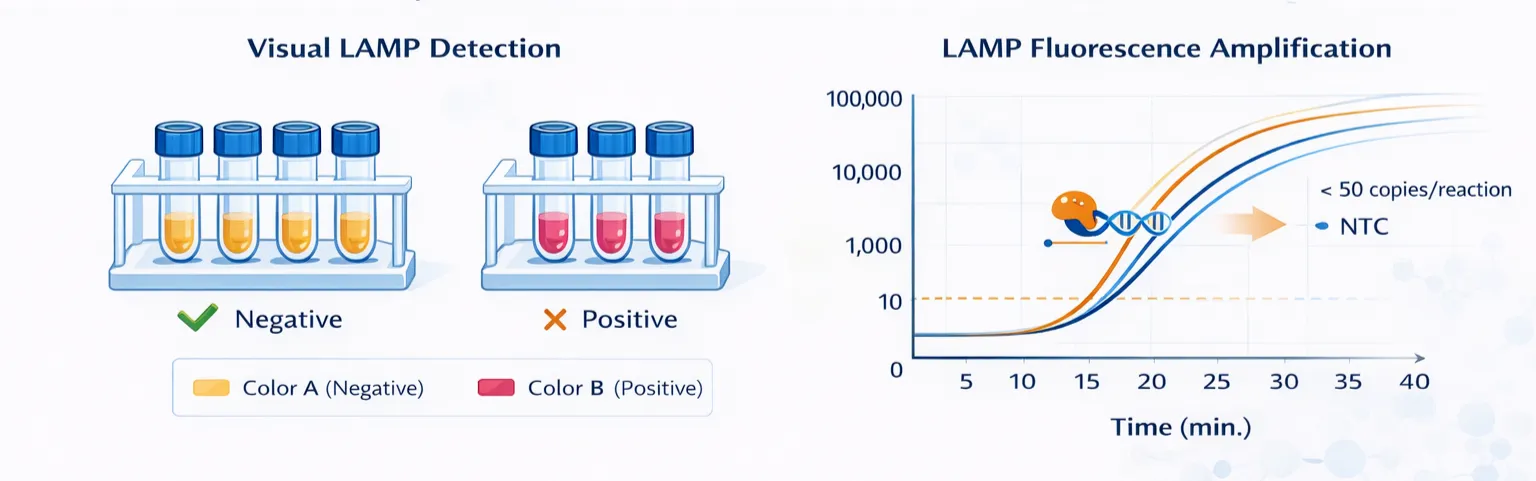
In parallel, a LAMP-based detection workflow was established to support rapid and equipment-light testing. The visual detection panel illustrates a clear color difference between negative and positive samples, demonstrating the feasibility of direct readout without complex instrumentation.
The fluorescence amplification profile further confirmed the analytical performance of the LAMP system. Positive reactions generated strong amplification signals within a short reaction window, while the negative control remained at baseline. The assay showed the capability to detect target input levels below 50 copies per reaction.
Companion diagnostics are most effective when developed in parallel with therapeutic agents.
We support drug-CDx co-development by enabling:
This integrated approach reduces development risk and enhances the probability of clinical success.
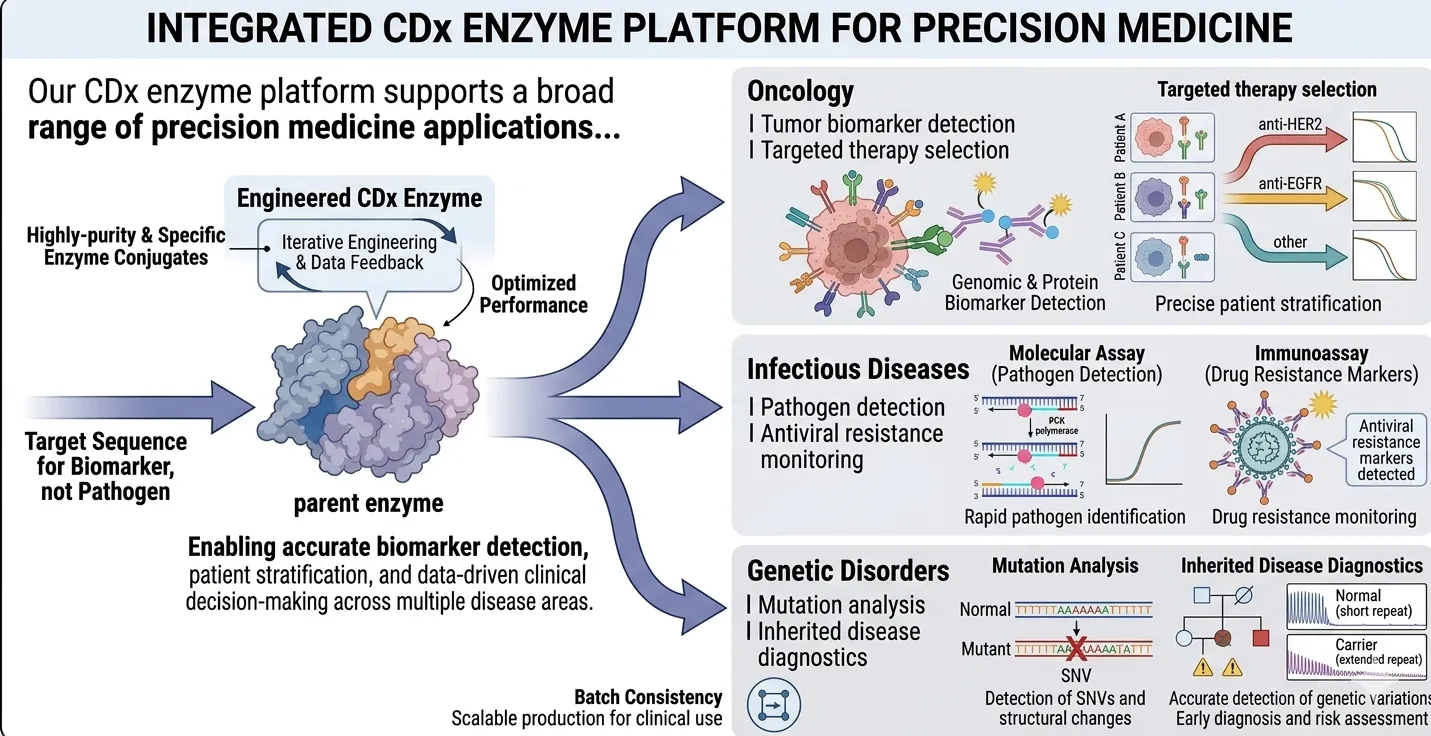
Our CDx enzyme platform supports a broad range of precision medicine applications, enabling accurate biomarker detection, patient stratification, and data-driven clinical decision-making across multiple disease areas.
Our platform supports detection of genomic and protein biomarkers associated with cancer progression and therapeutic response, enabling precise patient stratification and improved treatment outcomes in oncology CDx applications.
Enzyme-based molecular and immunoassay solutions enable rapid and sensitive detection of pathogens, as well as monitoring of drug resistance markers, supporting timely and effective infectious disease management.
High-sensitivity enzyme systems support accurate detection of genetic variations, including single nucleotide variants and structural changes, facilitating early diagnosis and risk assessment of inherited disorders.
Partner with Creative Enzymes to accelerate your companion diagnostic development.