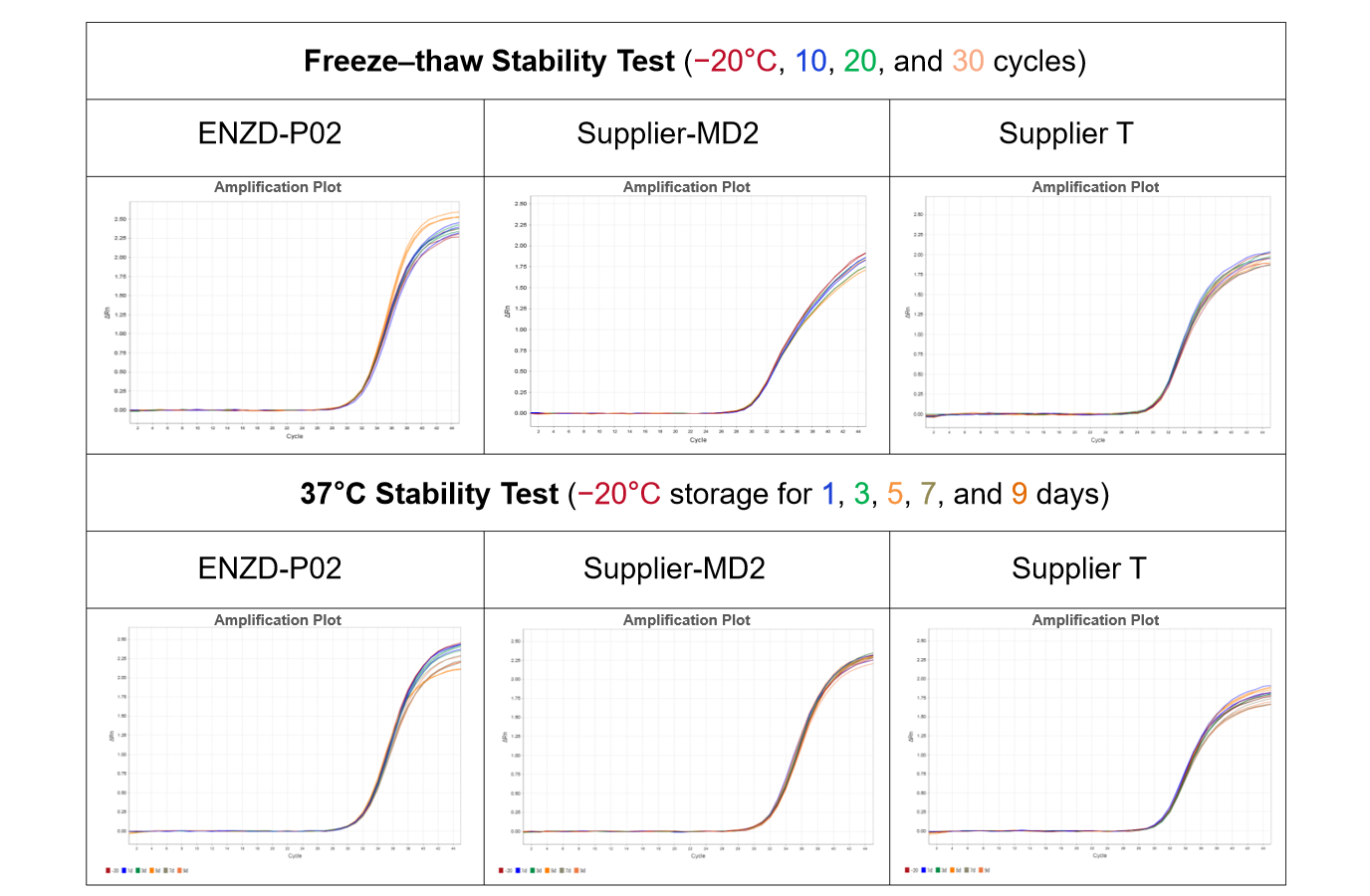
ENZD-P02 was evaluated alongside comparable products from three sources under identical qPCR conditions using the same primer–probe sets. The amplification profiles were compared in a singleplex system. The results demonstrated that ENZD-P02 achieved higher amplification sensitivity and a more stable plateau phase, indicating improved reaction efficiency and signal consistency. Data sourced from internal validation studies.
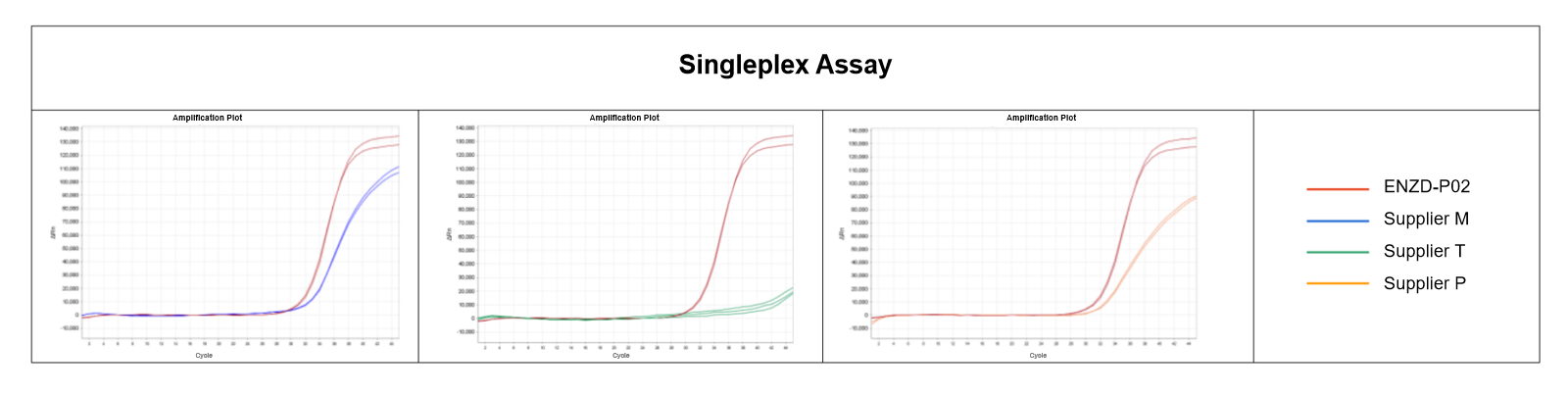
Multiplex qPCR experiments were conducted using the same sample to simultaneously amplify four target genes under identical conditions. Compared with a commercial reagent from Supplier T, ENZD-P02 showed superior plateau uniformity and amplification linearity across all targets, demonstrating robust performance in multiplex detection systems. Data sourced from internal validation studies.
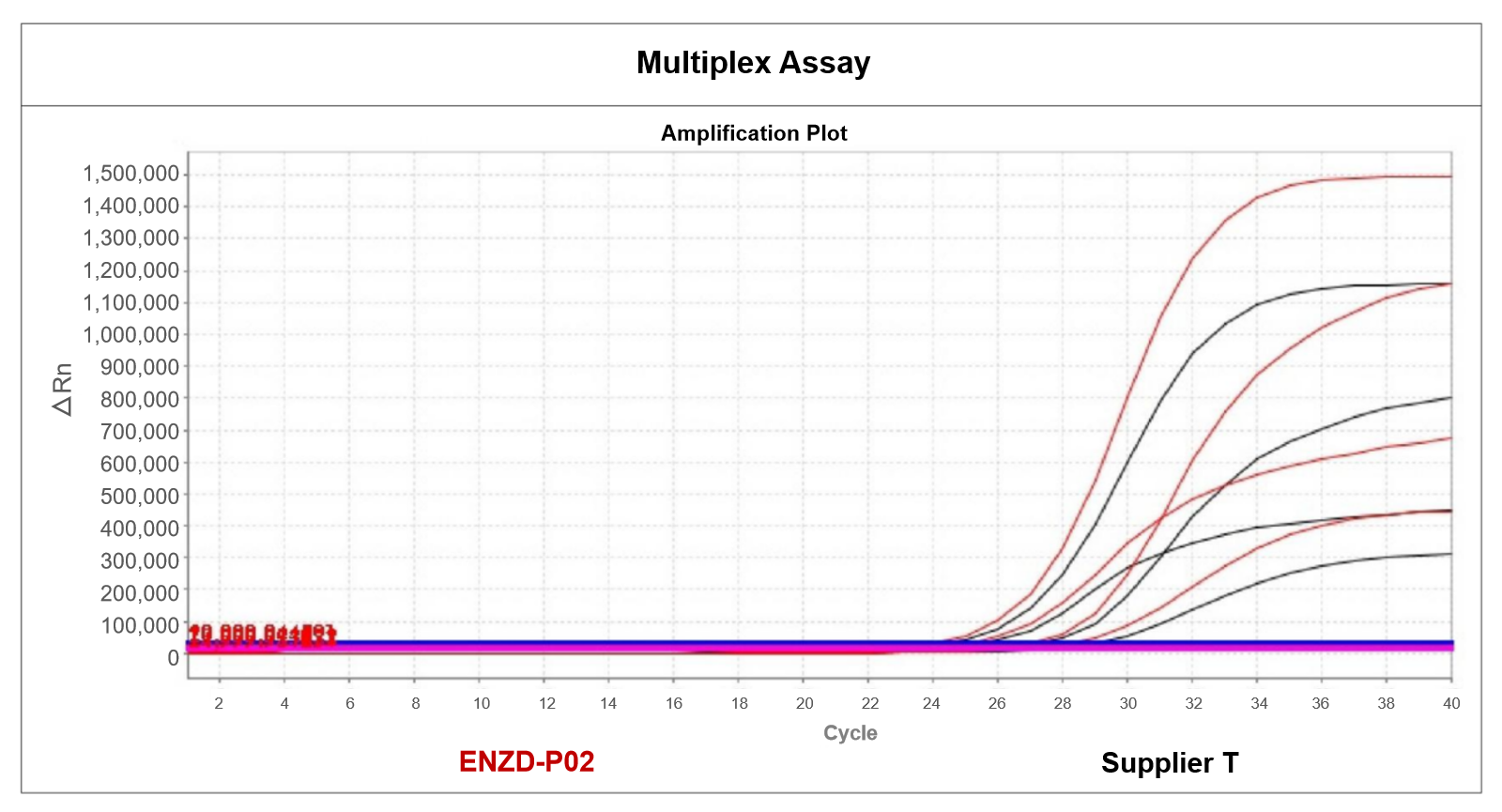
Detection performance was assessed across serial dilutions of template DNA. ENZD-P02 consistently achieved higher positive detection rates at low template concentrations compared to multiple commercial enzymes, while maintaining zero amplification in no-template controls (NTC). These results highlight its high sensitivity and robust discrimination capability, supporting accurate detection in low-copy-number scenarios. Data sourced from internal validation studies.
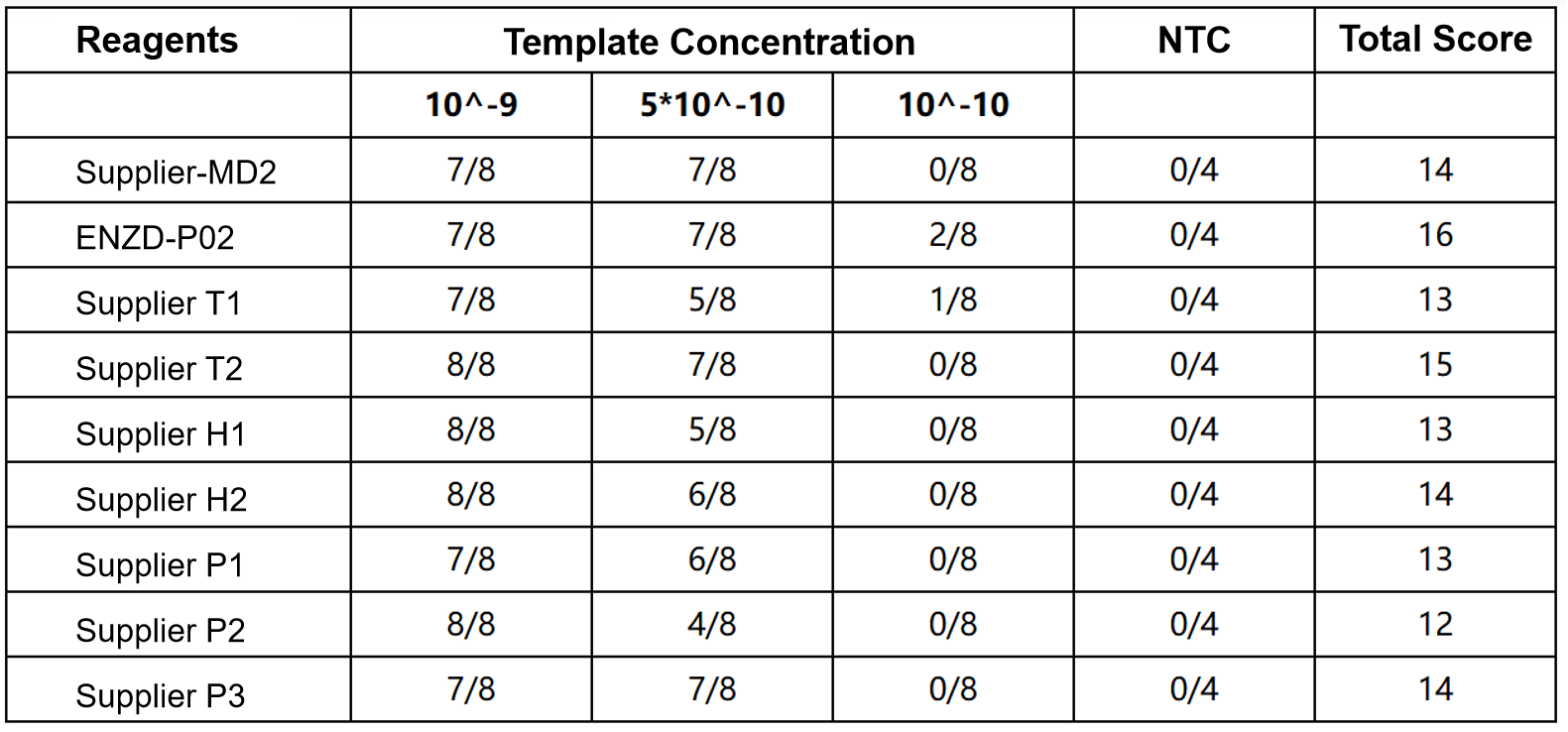
The stability of ENZD-P02 was evaluated under defined storage and operational conditions. The enzyme maintained consistent amplification performance over time, demonstrating reliable stability suitable for diagnostic workflow integration. Data sourced from internal validation studies.