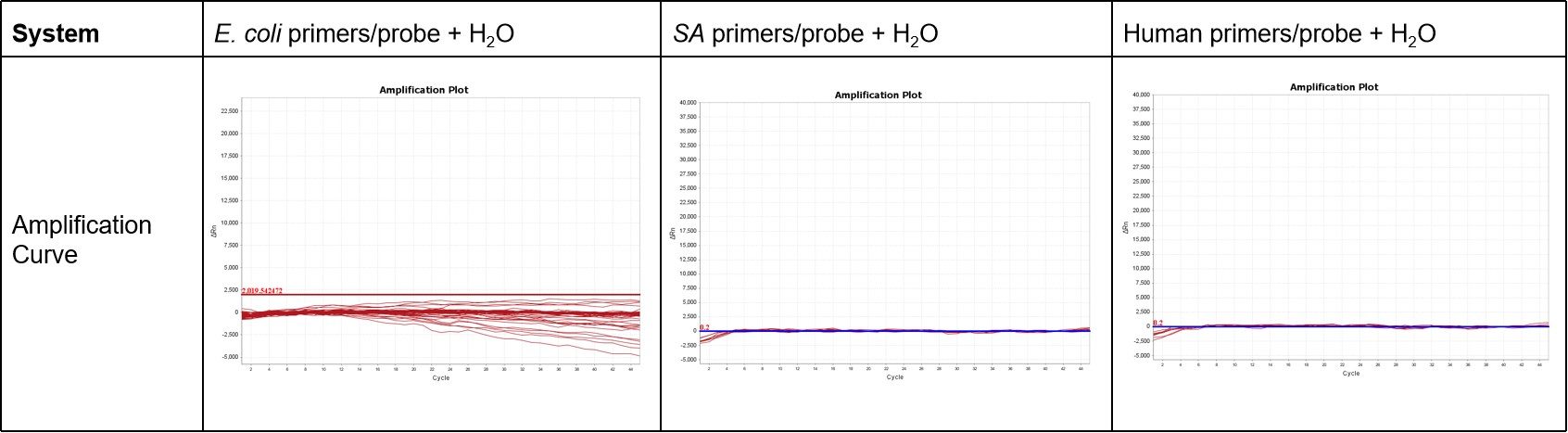
ENZD-QM01 was evaluated for residual host DNA contamination using no-template control (NTC) amplification across multiple detection systems, including E. coli, Staphylococcus aureus (SA), and human-specific primer–probe sets.
The results showed no detectable amplification in all NTC reactions (48 replicates), indicating extremely low levels of residual host DNA background in the reagent system and ensuring high assay reliability. These results are derived from internal validation studies evaluating residual host DNA levels across multiple detection systems.
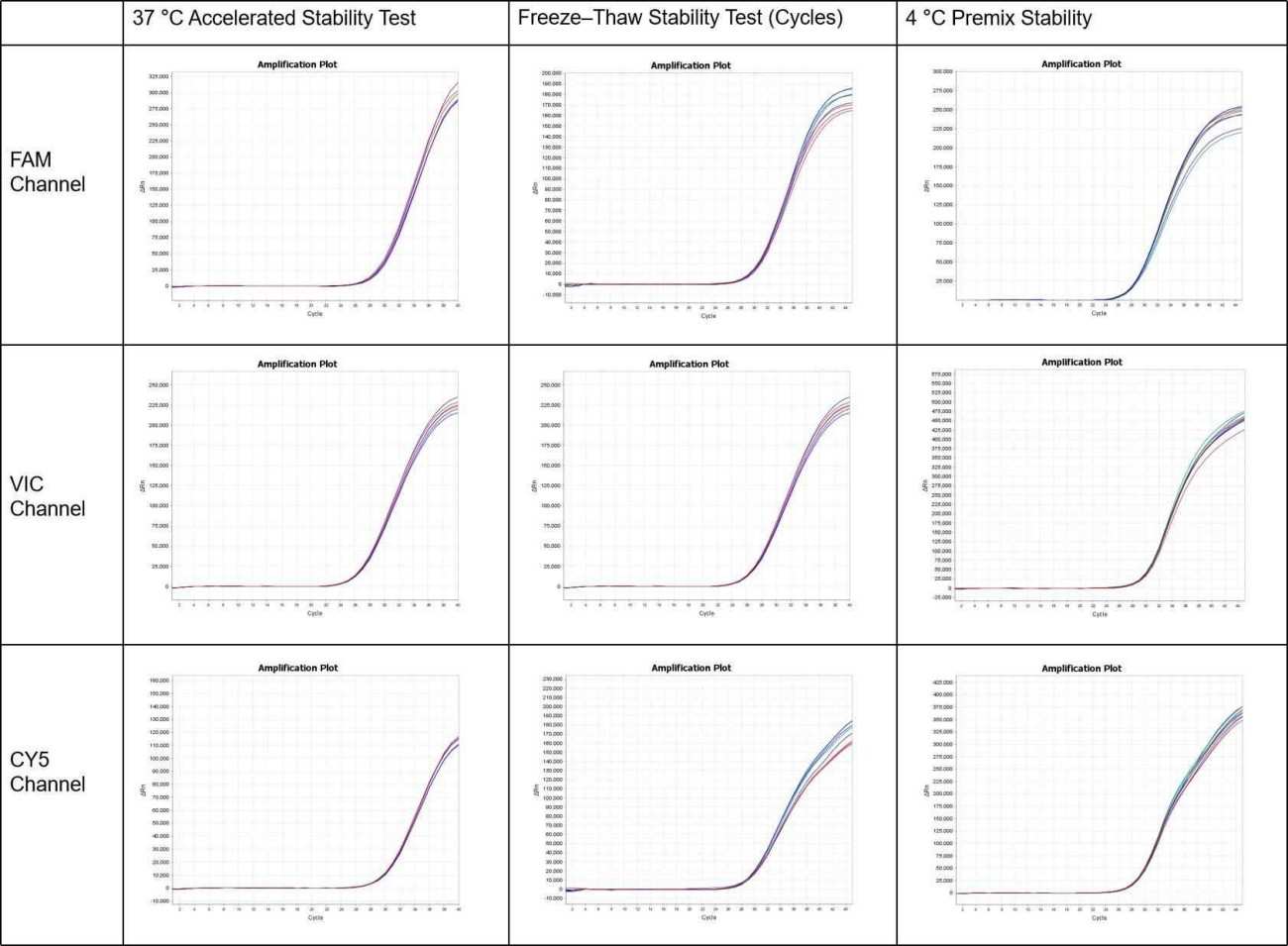
The premix stability of ENZD-QM01 was evaluated after pre-mixing with primers and probes under multiple stress conditions, including incubation at 37°C for 7 days, 60 freeze–thaw cycles, and storage at 4°C for up to 4 months.
Compared with the -20°C control group, no significant differences in amplification performance were observed across all tested conditions, demonstrating strong premix stability and robustness for storage and transport. These findings are based on internal validation studies assessing premix stability under thermal stress, freeze–thaw cycles, and extended storage conditions.
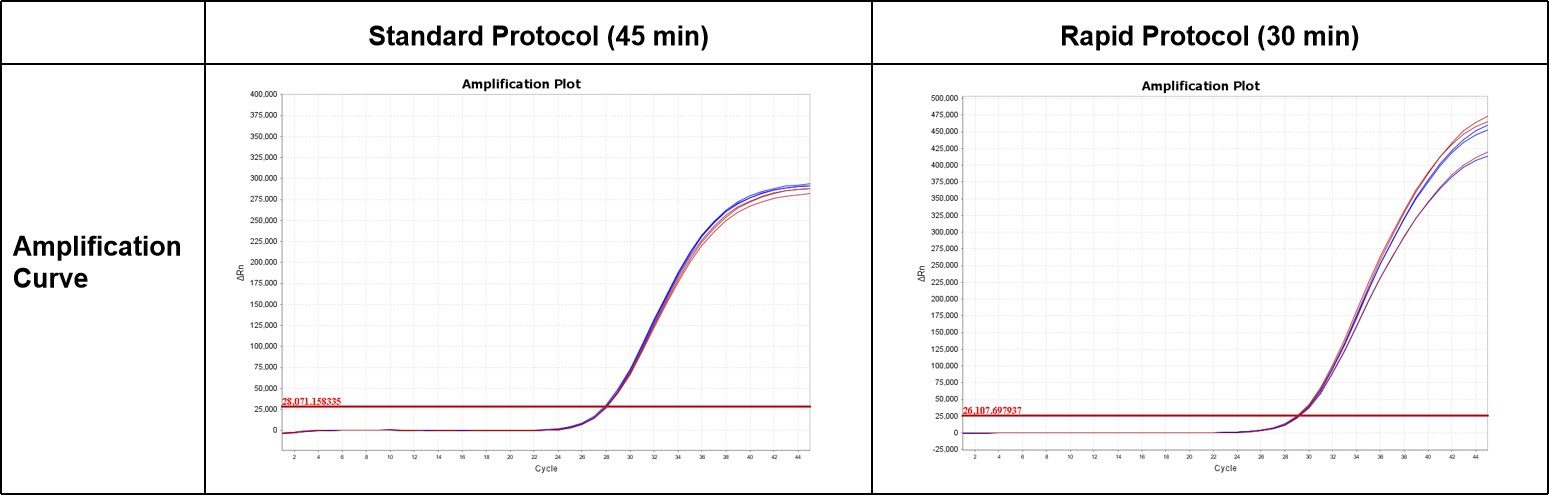
The performance of ENZD-QM01 was evaluated under both standard and rapid qPCR cycling protocols.
The results demonstrated comparable amplification efficiency between rapid and standard cycling conditions, indicating that ENZD-QM01 maintains stable reaction performance in accelerated workflows while supporting improved testing efficiency. These results are based on internal validation studies evaluating amplification performance under standard and rapid qPCR cycling conditions.